Bloggers: Trygve Andreassen, Senior engineer, and Siver Moestue, Associate professor,
The MR Cancer Group, Department of circulation and medical imaging
Scientists all over the world are strenuously pursuing new ways of killing cancer cells. They look for so called “drug targets” – proteins the cancer cells need for rapid and uncontrolled proliferation. In collaboration with Karolinska Institutet in Sweden, we have discovered such a protein called CHPT1. Based on this discovery we think it may be possible to develop new drugs for treating breast cancer.
A needle in a haystack
Before we can develop a new cancer drug, we must have some idea about what the drug should do. We have to identify a “drug target” – a protein the drug must recognize, bind to and alter the function of. It is not as simple as it might sound – we have about 20,000 different protein coding genes in our DNA, and it has been estimated that they can produce up to 100,000 different proteins (Savage, Nature 2015). These proteins communicate with each other in various ways, which means that we are trying to find a needle in a pretty big haystack.
Cancer drugs are often designed to block processes that are important for cell division, or the copying of DNA that happens prior to every cell division. In our research group, we are interested in the metabolism of cancer cells (in order to grow and divide fast, the cancer cells will reprogram their metabolism), and therefore we have searched for possible target proteins in the biochemical pathways.
In collaboration with Karolinska Institutet in Sweden, we started out with some known characteristics of breast cancer cells: They often grow faster in the presence of oestrogen; and they have an abnormally high turnover of the molecule choline. In order to study the choline turnover we apply a technique called MR-spectroscopy.
Main suspect: CHPT1
First we identified about 18,000 oestrogen binding domains in DNA, and found that these domains control the expression of about 2500 genes. Next we found how many of these genes that could be involved in choline turnover – hereby reducing the number of candidate genes to 19. By interpreting MR-spectra of cancer cells grown with and without oestrogen, we found which metabolic changes that could be derived from oestrogen stimulation. This allowed us to further reduce the number of proteins – and eventually we suspected the protein CHPT1 of being heavily involved in transferring the stimulating effect of oestrogen to the metabolic machinery of cancer cells.
To make sure that our findings are not only valid for cancer cells grown in the lab, we investigated tumour tissue from 70 breast cancer patients. Here we found higher expression of CHPT1 in tumour tissue compared to normal breast tissue. Even more interesting, we could see that tumours classified as oestrogen sensitive had significantly higher CHPT1 expression than the other tumours. This implies that oestrogen actually activates CHPT1 in breast cancer patients.
The next step in proving that CHPT1 is a relevant drug target was to show what happens if we switch off this gene. In the laboratory we have molecular tools which allow us to stop the production of the CHPT1-protein in cancer cells, and we found that cancer cells grew much slower when CHPT1 was switched off (Figure 1).
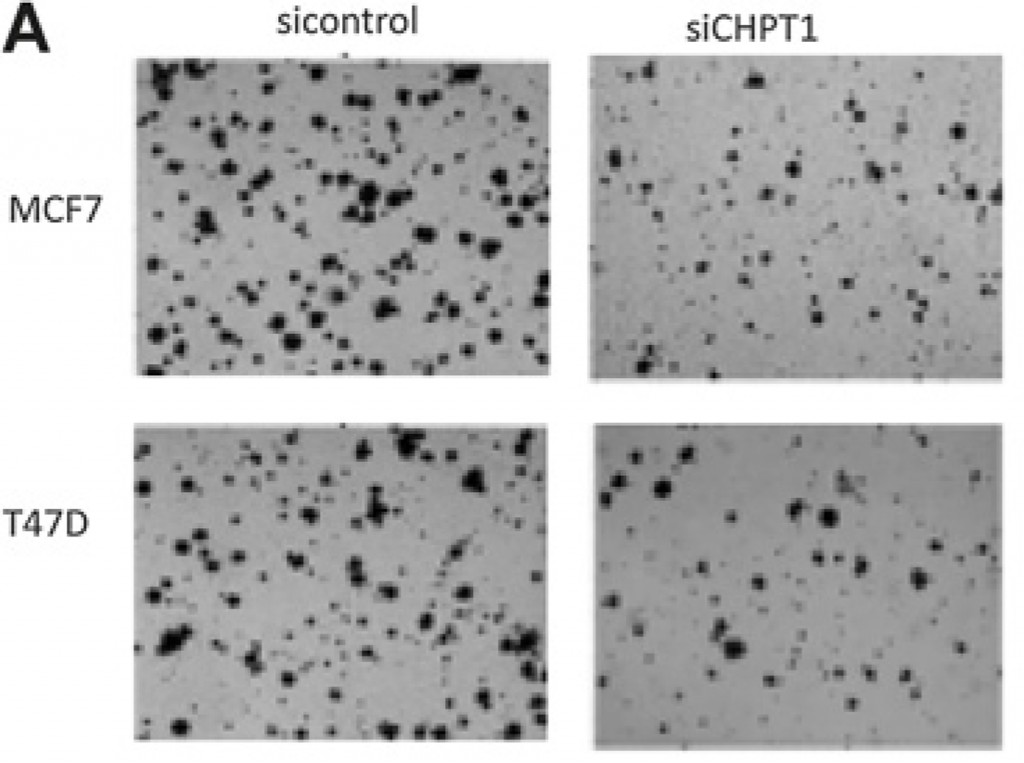
Figure 1: The two pictures to the left show to different types of breast cancer, seen in a microscope when growing freely in a dish. In the pictures to the right (marked siCHPT1) we see the same cells, but here we have “switched off” CHPT1. There are clearly fewer cells, which indicates that the cells need CHPT1 to grow and divide.
Killing cells in a dish at the lab is fairly easy, so the next step was to see if we could influence tumour growth in a living organism. Our choice of organism was zebrafish (they are quite transparent which enables us to observe the cancer cells directly). By switching off CHPT1 we could see that both tumour growth, as well as the cancer cells’ ability to spread to other tissue and organs, was reduced (Figure 2).
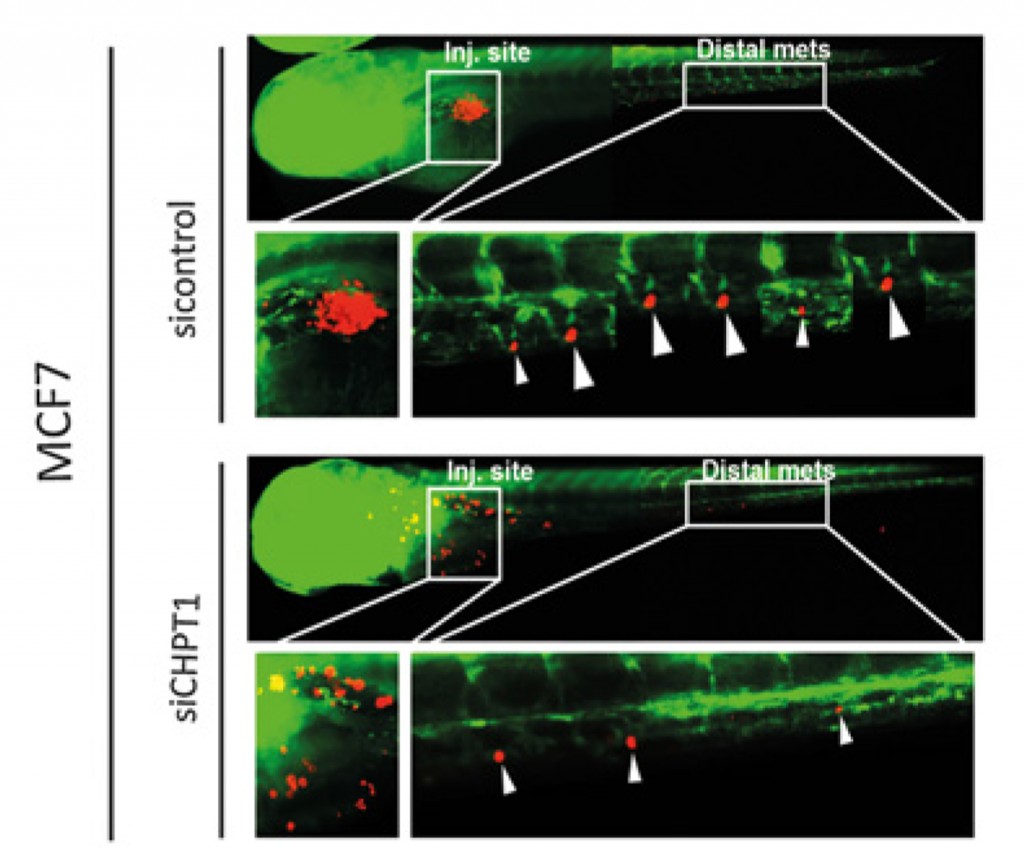
Figure 2: Above we see a zebrafish larva with implanted oestrogen sensitive MCF-7 cancer cells. They have developed a tumour at the injections site (red colour), and daughter tumours (metastases) have formed along the spine (marked with arrows (6). Below we see a zebrafish larva injected with the same type of cancer cells, but with CHPT1 switched off (siCHPT1). Here, there is no big tumour at the injection site, and there are clearly fewer daughter tumours along the spine (marked with arrows (3)).
All in all, we have presented several indicators suggesting that CHPT1 may be an important drug target for the treatment of oestrogen sensitive breast cancer. But there are still many things we haven’t found out yet: Is it possible to design drugs that block CHPT1? What are the side effects? Are they superior to existing drugs?
We have published our findings and hope that someone will take up the gauntlet and continue working on our ideas. And hopefully be able to develop a new drug based on our basic research.
The project has been partly funded by the Norwegian Cancer Society and the Research Council of Norway.

